Gas-phase basicity of cyclic guanidine derivatives – a DFT study†
Abstract
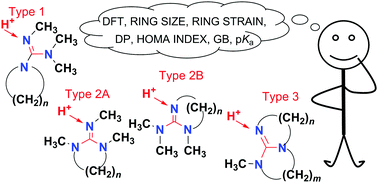
Density functional theory calculations (B3LYP) were employed in the study of gas-phase basicity (GB) and pKa of three different types of cyclic guanidines differing in the number of nitrogen atoms incorporated in rings. The large GB values were calculated for bigger ring sizes, and these guanidines fall within the superbases (GB > 240 kcal mol−1). Basicity is associated with ring strain (decreasing the GB effect with an increase in strain), which is the least pronounced in guanidines with only one nitrogen atom incorporated in the ring. Study of geometrical and electronic parameters (the degree of pyramidalization of nitrogen atoms (DP), s-character of the lone pair and HOMA index) provided more detailed information on the ring strain effects.


 Please wait while we load your content...
Please wait while we load your content...