Noble metal nanowire arrays as an ethanol oxidation electrocatalyst†
Abstract
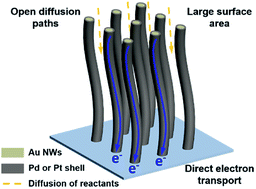
Vertically aligned noble metal nanowire arrays were grown on conductive electrodes based on a solution growth method. They show significant improvement of electrocatalytic activity in ethanol oxidation, from a re-deposited sample of the same detached nanowires. The unusual morphology provides open diffusion channels and direct charge transport pathways, in addition to the high electrochemically active surface from the ultrathin nanowires. Our best nanowire arrays exhibited much enhanced electrocatalytic activity, achieving a 38.0 fold increase in specific activity over that of commercial catalysts for ethanol electrooxidation. The structural design provides a new direction to enhance the electrocatalytic activity and reduce the size of electrodes for miniaturization of portable electrochemical devices.


 Please wait while we load your content...
Please wait while we load your content...