Uncovering the active sites and demonstrating stable catalyst for the cost-effective conversion of ethanol to 1-butanol†
Abstract
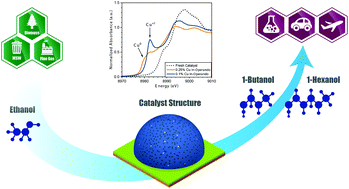
The recent emergence of a robust renewable ethanol industry has provided a sustainable platform molecule toward the production of value-added chemicals and fuels; what is lacking now are viable conversion processes from ethanol that can displace the current production pathways from non-renewable pathways. Here in the work, we demonstrate the highly selective conversion of ethanol to higher alcohols over low copper loaded MgAl mixed oxide catalysts, with 50% improvement in higher alcohol yields over the current state of the art. At these copper concentrations, atomically dispersed Cu1+ were found to be stable even at highly reductive conditions and highly active towards higher alcohol products (e.g. butanol, hexanol) while suppressing side reaction pathways and leading to extended lifetimes of over 150 hours time on stream. Technoeconomic analysis conducted based on these experimental results demonstrate that this catalytic system is cost-competitive with the conventional process. This marks significant progress in the development of Guerbet coupling of ethanol as a viable renewable process and offers a pathway toward sustainable chemical and fuel production.


 Please wait while we load your content...
Please wait while we load your content...