Emerging investigator series: rapid defluorination of 22 per- and polyfluoroalkyl substances in water using sulfite irradiated by medium-pressure UV†
Abstract
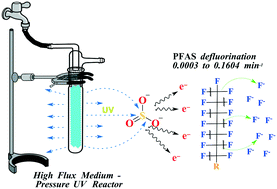
The removal of per- and polyfluoroalkyl substances (PFAS) from drinking water supplies is crucial to protect the public from their health hazards. We investigated rapid defluorination of 22 PFAS species using a high-photon-flux medium-pressure UV/sulfite process. Defluorination rates followed pseudo first-order kinetics ranging from 0.0003 to 0.1604 min−1. Defluorination was faster and less subject to the effects of pH and dissolved oxygen (O2) concentrations than lower energy systems, likely due to the greater number of hydrated electrons (eaq−) produced. Ammonium perfluoro-2-methyl-3-oxahexanoate (GenX) was the most rapidly defluorinated PFAS with a half-life of 4.3 min at pH 12 and 10 mM sulfite. Perfluorocarboxylic acids (PFCAs) also exhibited appreciable defluorination with half-lives between 7.8 and 577.6 min. PFCA defluorination rates increased with decreasing fluoroalkyl chain length. Perfluorooctanoic acid and perfluorooctanesulfonic acid, the most commonly detected PFAS in water, were rapidly defluorinated with half-lives of 11.3 and 22.1 min, respectively. Natural water constitutes present in surface and wastewater caused a decrease in defluorination efficiency. Overall, utilization of a medium-pressure lamp increased eaq− production and caused fast defluorination of selected PFAS. However, ∼97% of the energy produced by the medium-pressure lamp is not absorbed by sulfite, thus does not result in the production of eaq−, and is therefore wasted. The finding that the challenges of pH adjustment and scavenging by O2 may be overcome provides the pathway to field-scale applications (e.g., groundwater remediation) of either low-pressure or medium-pressure lamps given that the system energy input is great enough.
- This article is part of the themed collection: Emerging Investigator Series


 Please wait while we load your content...
Please wait while we load your content...