Natural organic matter adsorption conditions influence photocatalytic reaction pathways of phosphate-treated titanium dioxide nanoparticles†
Abstract
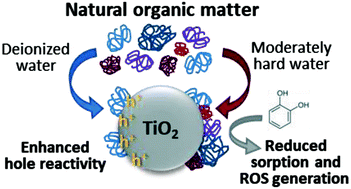
Titanium dioxide (TiO2) nanoparticles have been widely studied for water treatment applications; however, natural organic matter (NOM) is often reported to hamper the efficiency of the nanoparticles toward the degradation of target pollutants. Phosphate treatment has been proposed as a potentially facile solution to this problem, as phosphate competes for TiO2 surface sites to diminish the NOM adsorption. However, the potential importance of the conditions of the NOM exposure and the residual NOM remaining after phosphate treatment have not been fully explored. Here, we investigate the reactivity of phosphate-treated TiO2 nanoparticles with NOM coatings adsorbed from two background water chemistries, deionized water (TiO2–NOMDIW) and moderately hard water (TiO2–NOMMHW). Thorough characterization by size exclusion chromatography revealed that the adsorbed NOM was only partially displaced after phosphate treatment, with a higher adsorbed mass and wider variety of NOM species persisting on TiO2–NOMMHW compared to TiO2–NOMDIW. Although the remaining adsorbed NOM did not significantly influence the degradation rate of phenol as a model pollutant, remarkably distinct effects were observed in the degradation of catechol as an oxidative byproduct of phenol, with TiO2–NOMMHW hindering catechol degradation and TiO2–NOMDIW accelerating catechol degradation. The suppressed reactivity for TiO2–NOMMHW was attributed to hindrance of the physical adsorption of catechol to the TiO2 surface by the NOMMHW layer as well as changes in the reactive oxygen species profile as measured by electron paramagnetic resonance (EPR) spectroscopy, whereas the enhanced reactivity for TiO2–NOMDIW was attributed to higher hole formation, suggesting participation of the NOMDIW layer in electron transfer processes. This research highlights the critical importance of the NOM surface coating in directing the mechanisms for pollutant degradation in photocatalytic nano-enabled water treatment applications.


 Please wait while we load your content...
Please wait while we load your content...