Coulombically-stabilized oxygen hole polarons enable fully reversible oxygen redox†
Abstract
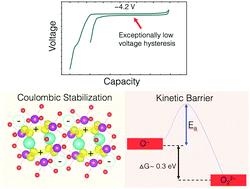
Stabilizing high-valent redox couples and exotic electronic states necessitates an understanding of the stabilization mechanism. In oxides, whether they are being considered for energy storage or computing, highly oxidized oxide-anion species rehybridize to form short covalent bonds and are related to significant local structural distortions. In intercalation oxide electrodes for batteries, while such reorganization partially stabilizes oxygen redox, it also gives rise to substantial hysteresis. In this work, we investigate oxygen redox in layered Na2−xMn3O7, a positive electrode material with ordered Mn vacancies. We prove that coulombic interactions between oxidized oxide-anions and the interlayer Na vacancies can disfavor rehybridization and stabilize hole polarons on oxygen (O−) at 4.2 V vs. Na/Na+. These coulombic interactions provide thermodynamic energy saving as large as O–O covalent bonding and enable ∼40 mV voltage hysteresis over multiple electrochemical cycles with negligible voltage fade. Our results establish a complete picture of redox energetics by highlighting the role of coulombic interactions across several atomic distances and suggest avenues to stabilize highly oxidized oxygen for applications in energy storage and beyond.
- This article is part of the themed collections: EES symposium collection and Energy Frontiers: Electrochemistry and Electrochemical Engineering


 Please wait while we load your content...
Please wait while we load your content...