Can sustainable ammonia synthesis pathways compete with fossil-fuel based Haber–Bosch processes?†
Abstract
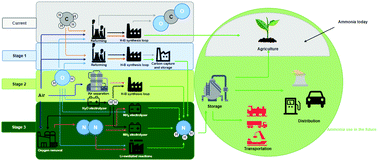
As renewable electricity prices continue to decline, interest grows in alternative routes for the synthesis of sustainable fuels and chemicals, including ammonia. Considering demand for fertilizers, as well as its future potential as a dispatchable energy vector, sustainable synthesis of ammonia is being explored as an alternative to the capital- and carbon-intensive fossil-fuel-driven Haber–Bosch process. Here we assess stages along a transition to the sustainable synthesis of ammonia, looking at economic feasibility and climate impacts compared to the incumbent Haber–Bosch without and with CO2 capture. This analysis enables us to suggest technological thresholds for sustainable synthesis of ammonia to become economically and environmentally favourable. When driven by renewable energy sources, the water electrolyzer (near $400 per kW) coupled Haber–Bosch process will reach cost parity near 2.5 cents per kWh electricity. In the case of direct electrochemical ammonia synthesis, achieving cost-parity using the same 2.5 cents per kWh electricity will rely on achieving major advances in performance: an electrolysis full-cell energy efficiency exceeding 40% at a current density of 0.5 A cm−2. Once this operating performance is reached, electrically-powered ammonia synthesis will bring climate benefits when coupled with low-carbon electricity (<180 gCO2e per kWh), achievable when over half of today's U.S. electricity generation is supplied by renewable energy sources. We conclude with a forward-looking perspective on the key challenges and opportunities for sustainable ammonia synthesis routes to be competitive with the incumbent Haber–Bosch process in the future.
- This article is part of the themed collection: SDG12: Sustainable production of energy materials


 Please wait while we load your content...
Please wait while we load your content...