Catalysis of CO2 reduction by diazapyridinophane complexes of Fe, Co, and Ni: CO2 binding triggered by combined frontier MO associations involving a SOMO†
Abstract
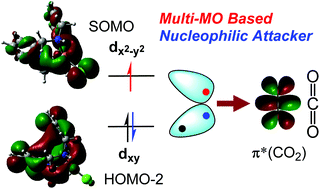
Our previous study on the photochemical CO2 reduction into CO catalyzed by the diazapyridinophane complexes of Fe, Co, and Ni revealed that (i) the Co catalyst shows the highest TOF but degrades rapidly, (ii) the Fe catalyst exhibits a lower TOF relative to Co but shows higher robustness, giving a higher TON, and (iii) the Ni complex shows no activity (Sakaguchi et al., Chem. Commun., 2019, 55, 8552). Here we show our DFT results unveiling that the Fe and Co catalysts can utilize multiple sets of frontier MO associations at the CO2 binding by including one of the SOMOs in a high-spin d7 Fe(I) and d8 Co(I) center, respectively, giving an increased driving force for these oxidative addition steps. Remarkably, two-electron reduction of CO2 to CO22− at the binding step is driven by the two electrons transferred from different d-based orbitals. The CoI species binds CO2 at the rate-limiting step with an activation barrier of 15.0 kcal mol−1, rationalizing the high initial TOF observed. However, the CoI(CO) species is given as a dead-end product, consistent with its relatively rapid deactivation. The Fe catalyst possesses a slightly higher barrier in CO2 binding (ΔG‡ = 15.8 kcal mol−1) but does not stabilize the FeI(CO) species which readily releases CO (ΔG = 3.5 kcal mol−1). The Ni catalyst has the smallest barrier in CO2 binding (ΔG‡ = 11.5 kcal mol−1) but the CO release is largely prohibited by the dead-end NiI(CO) species, consistent with its inactive character towards CO2 reduction. The combined results all satisfactorily explain the observed catalytic behaviors.


 Please wait while we load your content...
Please wait while we load your content...