Unexpected solution behaviour of ester-functionalized half-sandwich Ru(ii) and Ir(iii) complexes†
Abstract
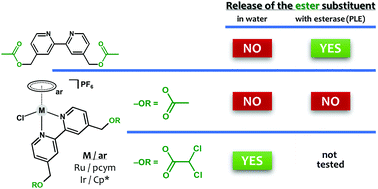
Complexes [Ru(η6-pcym)(bpydca)Cl]PF6 (Rudca) and [Ir(η5-Cp*)(bpydca)Cl]PF6 (Irdca) were developed as model compounds for the investigation of multi-targeted ester-functionalized half-sandwich ruthenium(II) and iridium(III) complexes; pcym = 1-methyl-4-(propan-2-yl)benzene (p-cymene), bpydca = 2,2′-bipyridine-4,4′-diyldimethanediyl bis(dichloroacetate), Cp* = pentamethylcyclopentadienyl. Aiming to understand the in-solution behaviour of these first-in-class complexes containing the pyruvate dehydrogenase kinase inhibitor dichloroacetate (dca) as the terminal bioactive substituent, several experiments were performed under aqueous conditions for Rudca and Irdca, as well as for compounds [Ru(η6-pcym)(bpyOH)Cl]PF6 (RuOH) and [Ir(η5-Cp*)(bpyOH)Cl]PF6 (IrOH), and acetyl analogues [Ru(η6-pcym)(bpyac)Cl]PF6 (Ruac) and [Ir(η5-Cp*)(bpyac)Cl]PF6 (Irac) bearing a different (biologically inactive) terminal substituent; bpyOH = 2,2′-bipyridine-4,4′-diyldimethanol, bpyac = 2,2′-bipyridine-4,4′-diyldimethanediyl diacetate. The experiments were also conducted in the presence of porcine liver esterase (PLE). All the six complexes were characterized by relevant techniques (e.g., NMR and mass spectrometry), including a single-crystal X-ray analysis of complexes Rudca, Ruac, RuOH and IrOH. Although designed as model compounds, Rudca, Irdca, RuOH and IrOH were also screened for their antiproliferative activity in four human cancer cell lines (HCT116 colon carcinoma, MDA-MB-231 and MCF-7 breast adenocarcinomas, DU145 prostate carcinoma), where the tested complexes did not show any effect (IC50 > 100 μM).


 Please wait while we load your content...
Please wait while we load your content...