Abstract
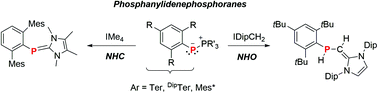
Phospha–Wittig reagents, RPPMe3 (R = Mes* 2,4,6-tBu3-C6H2; MesTer 2,6-(2,4,6-Me3C6H2)-C6H3; DipTer 2,6-(2,6-iPr2C6H3)-C6H3), can be considered as phosphine-stabilized phosphinidenes. In this study we show that PMe3 can be displaced by NHCs or NHOs. Interestingly, phosphinidene-like reactivity results in a subsequent C(sp2)–H activation of the exocyclic CH2 group in NHOs. This concept was further extended to allyl-apended NHOs, which resulted in phosphine-substituted allyl species.
- This article is part of the themed collection: #RSCPoster Conference


 Please wait while we load your content...
Please wait while we load your content...