Early-stage particle fragmentation behavior of a commercial silica-supported metallocene catalyst†
Abstract
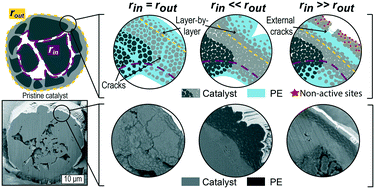
Efficient fragmentation of catalyst particles during the polymerization of olefins is a necessary process to maintain catalyst activity and obtain discrete polymer particles with ideal density and morphology. Collecting experimental evidence on the fragmentation process is essential for a fundamental understanding of this phenomenon. However, visualizing the very early onset of fragmentation within catalyst particles has been proven challenging. This study investigates the development of fragmentation at different reaction conditions in an industrial SiO2-supported metallocene catalyst of the type Zr/MAO/SiO2 (MAO = methylaluminoxane) early-stage of ethylene polymerization, i.e., in the range 1–20 g polyethylene per g catalyst. Focused ion beam-scanning electron microscopy was employed to visualize particle cross-sections with high 2-D resolution (i.e., a few tens of nm), showing that ethylene polymerization starts at two main fronts: one at the outer surface and one at the walls of the macropores within the particle. The relative rate of polymerization at these fronts was assessed by estimating the extent of polyethylene, catalyst, and macroporosity within each cross-section via image segmentation. When polymerization at these fronts proceeds at similar rates, fragmentation is extensive and a combination of both layer-by-layer and bi-sectioning modes; conversely, an imbalance between the two due to unideal reaction conditions, non-homogeneous active site distribution, or insufficient macroporosity leads to a sub-optimal fragmentation. This methodology revealed a detailed understanding of catalyst fragmentation for this commercial catalyst. However, it can be applied to a broader range of polymerization catalysts and parameters, amplifying the knowledge on such an intricated phenomenon.
- This article is part of the themed collection: Catalysis Science & Technology 10th Anniversary Symposium


 Please wait while we load your content...
Please wait while we load your content...