Mechanistic understanding of methane-to-methanol conversion on graphene-stabilized single-atom iron centers†
Abstract
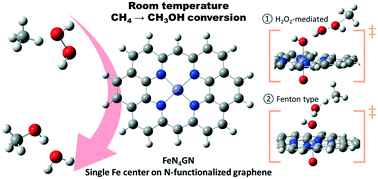
The functionalization of methane to value-added liquid chemicals remains as one of the “grand challenges” in chemistry. In this work, we provide insights into the direct methane-to-methanol conversion mechanisms with H2O2 as an oxidant on single Fe-atom centers stabilized on N-functionalized graphene, using first principles calculations. By investigating a series of different reaction paths on various active centers and calculating their turnover frequencies, we reveal that a H2O2-mediated radical mechanism and a Fenton-type mechanism are energetically the most plausible pathways taking place on di- and mono-oxo centers, respectively. Due to the thermodynamic preference of the mono-oxo center formation over the di-oxo under reaction conditions, the Fenton-type mechanism appears to determine the overall catalytic activity. On the other hand, the hydroxy(oxo) center, which is thermodynamically the most favorable center, is found to be catalytically inactive. Hence, the high activity is attributed to a fine balance of keeping the active centers as oxo-species during the reaction. Moreover, we reveal that the presence of solvent (water) can accelerate or slow down different pathways with the overall turnover of the dominant Fenton-type reaction being decreased. Importantly, this work reveals the nature of active sites and a gamut of reaction mechanisms for the direct conversion of methane to methanol rationalizing experimental observations and aiding the search for room temperature catalysts for methane conversion to liquid products.


 Please wait while we load your content...
Please wait while we load your content...