Simultaneous screening of multiple substrates with an unspecific peroxygenase enabled modified alkane and alkene oxyfunctionalisations†
Abstract
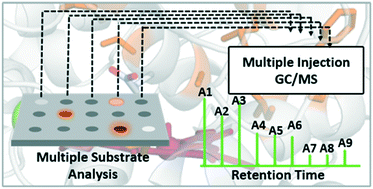
A high throughput GC-MS approach was developed, permitting the simultaneous analysis of up to three substrates and six products quantitatively from one reaction mixture. This screening approach was applied to site-saturation libraries of the novel unspecific peroxygenase MthUPO. Using this setup enabled substantial insights from a small mutant library. Enzyme variants were identified exhibiting selective alkene epoxidation and substantially shifted regioselectivities to 2- and 1-octanol formations. Computational modelling rationalised the observed selectivity changes.


 Please wait while we load your content...
Please wait while we load your content...