Late-stage difluoromethylation: concepts, developments and perspective
Abstract
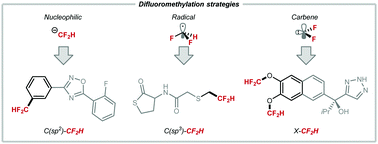
This review describes the recent advances made in difluoromethylation processes based on X–CF2H bond formation where X is C(sp), C(sp2), C(sp3), O, N or S, a field of research that has benefited from the invention of multiple difluoromethylation reagents. The last decade has witnessed an upsurge of metal-based methods that can transfer CF2H to C(sp2) sites both in stoichiometric and catalytic mode. Difluoromethylation of C(sp2)–H bond has also been accomplished through Minisci-type radical chemistry, a strategy best applied to heteroaromatics. Examples of electrophilic, nucleophilic, radical and cross-coupling methods have appeared to construct C(sp3)–CF2H bonds, but cases of stereoselective difluoromethylation are still limited. In this sub-field, an exciting departure is the precise site-selective installation of CF2H onto large biomolecules such as proteins. The formation of X–CF2H bond where X is oxygen, nitrogen or sulfur is conventionally achieved upon reaction with ClCF2H; more recently, numerous protocols have achieved X–H insertion with novel non-ozone depleting difluorocarbene reagents. All together, these advances have streamlined access to molecules of pharmaceutical relevance, and generated interest for process chemistry.


 Please wait while we load your content...
Please wait while we load your content...