Femtosecond intersystem crossing to the reactive triplet state of the 2,6-dithiopurine skin cancer photosensitizer†
Abstract
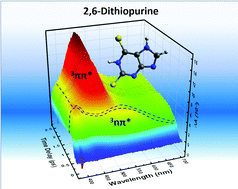
Site-selected sulfur-substituted nucleobases are a class of all organic, heavy-atom-free photosensitizers for photodynamic therapy applications that exhibit excellent photophysical properties such as strong absorption in the ultraviolet-A region of the electromagnetic spectrum, near-unity triplet yields, and a high yield of singlet oxygen generation. Recent investigations on doubly thionated nucleobases, 2,4-dithiothymine, 2,4-dithiouracil, and 2,6-dithiopurine, demonstrated that these set of dithionated nucleobases outperform the photodynamic efficacy exhibit by 4-thiothymidine–the most widely studied singly substituted thiobase to date. Out of the three dithionated nucleobases, 2,6-dithiopurine was shown to be the most effective, exhibiting inhibition of cell proliferation of up to 63% when combined with a low UVA dose of 5 J cm−2. In this study, we elucidated the electronic relaxation pathways leading to the population of the reactive triplet state of 2,6-dithiopurine. 2,6-Dithiopurine populates the triplet manifold in less than 150 fs, reaching the nπ* triplet state minimum within a lifetime of 280 ± 50 fs. Subsequently, the population in the nπ* triplet state minimum internally converts to the long-lived ππ* triplet state within a lifetime of 3 ± 1 ps. The relatively slow internal conversion lifetime is associated with major conformational relaxation in going from the nπ* to ππ* triplet state minimum. A unity triplet yield of 1.0 ± 0.1 is measured.
- This article is part of the themed collection: 2021 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...