Hydrogen solution in high-entropy alloys†
Abstract
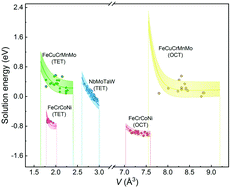
High-entropy alloys (HEAs) have been reported to have superior ability in hydrogen (H) storage and strong resistance to H embrittlement. These exceptional properties are directly related to the H solution in the HEAs. However, the diversity of atomic environments in the HEAs complicate the calculation of the H solution energy. With regard to this, we clarified an origin causing the variety of solution energy from the viewpoint of chemical and elastic interactions of H with the host atoms. Combining the semi-empirical atom potential and first-principles calculations regarding H in FeCrCoNi, NbMoTaW, and FeCuCrMnMo, we found that the elastic interaction presents a visibly linear relationship with the volume expansion caused by H insertion. By contrast, the chemical interaction shows a non-linear relationship with the volume of the interstitial polyhedron. A universal model was then established to generalize the solution energy of H. This model can expeditiously assess the H distribution and provide insight into evolution of the microstructure in HEAs.


 Please wait while we load your content...
Please wait while we load your content...