The kinetics of metal soap crystallization in oil polymers†
Abstract
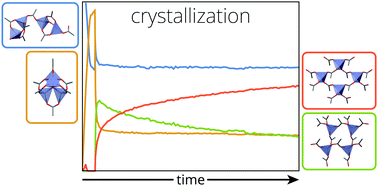
The crystallization of metal soaps in oil paint is an important chemical phenomenon that affects the appearance and structural stability of many works of art. A deep understanding of the structural transitions that occur during crystallization and their kinetics will help to support conservation decisions that minimize future detrimental change to paintings. We have used a method based on attenuated total reflection Fourier transform infrared spectroscopy and detailed spectrum analysis to quantitatively monitor all relevant metal soap structures during crystallization in a linseed oil matrix with varying degrees of polymerization. It was found that zinc soap crystallization behaviour is strongly influenced by the properties of the oil matrix, slowing down drastically with increasing polymerization, forming crystalline polymorphs in varying ratios, and demonstrating two-stage kinetics. In contrast, lead soap crystallization was invariably fast, but the degree of disorder in the crystallized phases was increasing with matrix polymerization. Besides fundamental insight into the mechanisms of metal soap crystallization, the results lay foundations for improved risk assessment during conservation treatment of oil paintings.


 Please wait while we load your content...
Please wait while we load your content...