Subtle hydrogen bonds: benchmarking with OH stretching fundamentals of vicinal diols in the gas phase†
Abstract
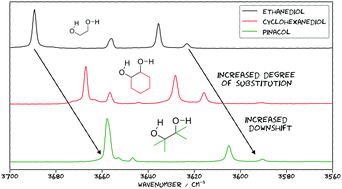
The theoretical description of spectral signatures for weakly bound hydrogen contacts between alcohol groups is challenging and remains poorly characterised. By combining Raman jet spectroscopy with appropriately scaled harmonic DFT predictions and relaxation path analyses for 16 vicinal diols (ethylene glycol (ethane-1,2-diol), propane-1,2-diol, 3,3,3-trifluoro-propane-1,2-diol, rac-butane-2,3-diol, 2-methyl-propane-1,2-diol, 2-methyl-butane-2,3-diol, pinacol (2,3-dimethyl-butane-2,3-diol), 3-butene-1,2-diol, 1-phenyl-ethane-1,2-diol, trans-cyclobutane-1,2-diol, trans-cyclopentane-1,2-diol, trans-cyclohexane-1,2-diol, trans-cycloheptane-1,2-diol, cis-cyclohexane-1,2-diol, 1-(1-hydroxy-1-methylethyl)-cyclopentanol and [1,1′-bicyclopentyl]-1,1′-diol), 69 conformational assignments become possible in a two-tier approach with a 5 diol training and an 11 diol test set. The latter reveals systematic deviations for ring strain and secondary π interactions, but otherwise a remarkably robust correction and correlation model based on hybrid DFT with a minimally augmented triple-zeta basis set is obtained, whereas GGA functionals perform significantly worse. Raw experimental data in the 3560–3700 cm−1 wavenumber range as well as computed geometries of all conformations invite further vibrational and structural benchmarking at the onset of hydrogen bonding. Beyond this diol-probed threshold, the accurate prediction of hydrogen bond induced shifts of different magnitudes remains one of the challenges for DFT functionals.
- This article is part of the themed collection: 2021 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...