Electron attachment to isolated and microhydrated favipiravir†
Abstract
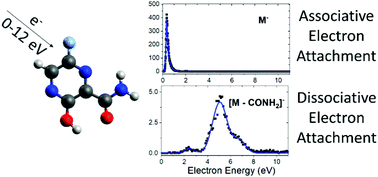
Electron attachment and its equivalent in complex environments, single-electron reduction, are important in many biological processes. Here, we experimentally study the electron attachment to favipiravir, a well-known antiviral agent. Electron attachment spectroscopy is used to explore the energetics of associative (AEA) and dissociative (DEA) electron attachment to isolated favipiravir. AEA dominates the interaction and the yields of the fragment anions after DEA are an order of magnitude lower than that of the parent anion. DEA primary proceeds via decomposition of the CONH2 functional group, which is supported by reaction threshold calculations using ab initio methods. Mass spectrometry of small favipiravir–water clusters demonstrates that a lot of energy is transferred to the solvent upon electron attachment. The energy gained upon electron attachment, and the high stability of the parent anion were previously suggested as important properties for the action of several electron-affinic radiosensitizers. If any of these mechanisms cause synergism in chemo-radiation therapy, favipiravir could be repurposed as a radiosensitizer.
- This article is part of the themed collection: PCCP 2021 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...