Impact of tensile and compressive forces on the hydrolysis of cellulose and chitin†
Abstract
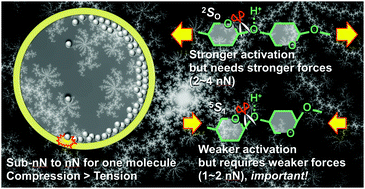
Mechanochemistry enables unique reaction pathways in comparison to conventional thermal reactions. Notably, it can achieve selective hydrolysis of cellulose and chitin, a set of abundant and recalcitrant biomass, by solvent-free ball-milling in the presence of acid catalysts. Although the merits of mechanochemistry for this reaction are known, the reaction mechanism is still unclear. Here, we show how the mechanical forces produced by ball-milling activate the glycosidic bonds of carbohydrate molecules towards hydrolysis. This work uses experimental and theoretical evaluations to clarify the mechanism. The experimental results reveal that the ball-mill accelerates the hydrolysis by mechanical forces rather than local heat. Meanwhile, the classical and quantum mechanics calculations indicate the subnano to nano Newton order of tensile and compressive forces that activate polysaccharide molecules in the ball-milling process. Although previous studies have taken into account only the stretching of the molecules, our results show that compressive forces are stronger and effective for the activation of glycosidic bonds. Accordingly, in addition to stretching, compression is crucial for the mechanocatalytic reaction. Our work connects the classical physics of ball-milling on a macro scale with molecular activation at a quantum level, which would help to understand and control mechanochemical reactions.


 Please wait while we load your content...
Please wait while we load your content...