Dynamical theory for the battery's electromotive force
Abstract
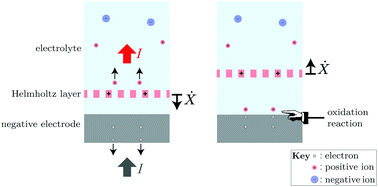
We propose a dynamical theory of how the chemical energy stored in a battery generates the electromotive force (emf). In this picture, the battery's half-cell acts as an engine, cyclically extracting work from its underlying chemical disequilibrium. We show that the double layer at the electrode–electrolyte interface can exhibit a rapid self-oscillation that pumps an electric current, thus accounting for the persistent conversion of chemical energy into electrical work equal to the emf times the separated charge. We suggest a connection between this mechanism and the slow self-oscillations observed in various electrochemical cells, including batteries, as well as the enhancement of the current observed when ultrasound is applied to the half-cell. Finally, we propose more direct experimental tests of the predictions of this dynamical theory.


 Please wait while we load your content...
Please wait while we load your content...