Non-adiabatic quantum interference in the ultracold Li + LiNa → Li2 + Na reaction
Abstract
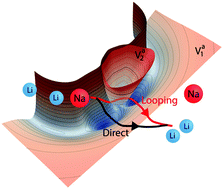
Electronically non-adiabatic effects play an important role in many chemical reactions. However, how these effects manifest in cold and ultracold chemistry remains largely unexplored. Here for the first time we present from first principles the non-adiabatic quantum dynamics of the reactive scattering between ultracold alkali-metal LiNa molecules and Li atoms. We show that non-adiabatic dynamics induces quantum interference effects that dramatically alter the ultracold rotationally resolved reaction rate coefficients. The interference effect arises from the conical intersection between the ground and an excited electronic state that is energetically accessible even for ultracold collisions. These unique interference effects might be exploited for quantum control applications such as a quantum molecular switch. The non-adiabatic dynamics are based on full-dimensional ab initio potential energy surfaces for the two electronic states that includes the non-adiabatic couplings and an accurate treatment of the long-range interactions. A statistical analysis of rotational populations of the Li2 product reveals a Poisson distribution implying the underlying classical dynamics are chaotic. The Poisson distribution is robust and amenable to experimental verification and appears to be a universal property of ultracold reactions involving alkali metal dimers.
- This article is part of the themed collection: 2021 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...