Blue- and white-light-emitting 2D-coordination polymers and their solid-state photodimerization reaction†
Abstract
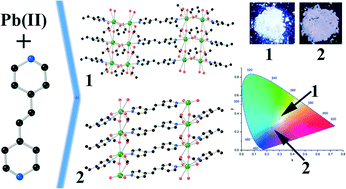
Two Pb(II) coordination polymers (CPs), namely [Pb(μ-bpe)(μ-O2CCH3)2]n (1) and [Pb(μ-bpe)(μ-O2CH)2]n (2), have been isolated from the reaction of Pb(OAc)2·3H2O with 1,2-bis(4-pyridyl)ethene (bpe) in the presence of acetate and formate anionic ligands, respectively. Compounds 1 and 2 formed 2D sheet-like structures, constructed by bridging acetate and formate carboxylate ligands. The bpe ligand containing the olefinic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds was perfectly brought to the proximity of Schmidt topolochemical criteria for 2. The 2D sheet in 2 underwent 67% of the photodimerization reaction in the solid state, while the 2D sheet in 1 was photochemically stable. Therefore, the photochemical reactivity of 1 and 2 was rationalized based on the orientation of the olefinic C
C bonds was perfectly brought to the proximity of Schmidt topolochemical criteria for 2. The 2D sheet in 2 underwent 67% of the photodimerization reaction in the solid state, while the 2D sheet in 1 was photochemically stable. Therefore, the photochemical reactivity of 1 and 2 was rationalized based on the orientation of the olefinic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds, which was brought close to each other due to the distinct bridging of the carboxylates and the Pb(II) metal ions. The room temperature solid-state photoluminescence (PL) revealed that compound 1 exhibited a near white-light emission and the blue-light emission for 2 upon excitation at 365 nm. The differences in the emission of the two 2D-sheets were correlated with the bridging carboxylate ligands.
C bonds, which was brought close to each other due to the distinct bridging of the carboxylates and the Pb(II) metal ions. The room temperature solid-state photoluminescence (PL) revealed that compound 1 exhibited a near white-light emission and the blue-light emission for 2 upon excitation at 365 nm. The differences in the emission of the two 2D-sheets were correlated with the bridging carboxylate ligands.
- This article is part of the themed collection: Coordination Networks


 Please wait while we load your content...
Please wait while we load your content...