Use of nitrogen-doped amorphous carbon nanodots (N-CNDs) as a fluorometric paper-based sensor: a new approach for sensitive determination of lead(ii) at a trace level in highly ionic matrices†
Abstract
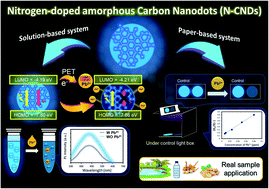
This work reports a facile synthesis of nitrogen-doped amorphous carbon nanodots (N-CNDs) and their use as a fluorometric paper-based sensor for the determination of Pb2+ at a low concentration. Both solution-based and paper-based systems were developed. The results show that the linearity ranges for Pb2+ determination were 0.010–10 mg L−1 (LOD = 0.008 mg L−1) and 0.005–0.075 mg L−1 (LOD = 0.004 mg L−1) for the solution-based and the paper-based sensors, respectively. Furthermore, the developed sensors show relatively high selectivity toward Pb2+ over ten other metal cations of different charges including As3+, Hg2+, Cd2+, Mg2+, Ni2+, Zn2+, Fe3+, Cu2+, Ba2+, and Ag+. The mechanism of Pb2+ determination was also investigated. It was found that the sensors exploited the quenching of the fluorescence intensity of N-CNDs by Pb2+via the photo-induced electron transfer (PET) mechanism. When applied to real water and herbal medicine samples, the performance of the sensor exhibited no significant difference as compared to the results of the validation method (ICP-OES). Overall, the developed sensors, especially the paper-based one, are promising for the practical analysis of Pb2+ in pharmaceutical and environmental samples with a low Pb2+ concentration.


 Please wait while we load your content...
Please wait while we load your content...