Plug-and-play assembly of paper-based colorimetric and electrochemical devices for multiplexed detection of metals†
Abstract
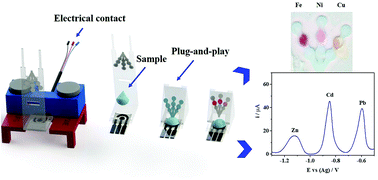
Heavy metals are the main pollutants present in aquatic environments and their presence in human organisms can lead to many different diseases. While many methods exist for analysis, colorimetric and electrochemistry are particularly attractive for on-site analysis and their integration on a single platform can improve multiplexed metals analysis. This report describes for the first time a “plug-and-play” (PnP) assembly for coupling a microfluidic paper-based device (μPAD) and a screen-printed electrochemical paper-based device (ePAD) using a vertical and reversible foldable mechanism for multiplexed detection of Fe, Ni, Cu, Zn, Cd and Pb in river water samples. The integration strategy was based on a reversible assembly, allowing the insertion of a pretreatment zone to minimize potential chemical interfering agents and providing a better control of the aspirated sample volume as well as to a lower sample evaporation rate. In comparison with lateral flow and electrochemical assays performed using independent devices, the integrated prototype proved that the reversible coupling mechanism does not interfere on the analytical performance (95% confidence interval). The limit of detection (LOD) values calculated for metals determined varied from 0.1 to 0.3 mg L−1 (colorimetric) and from 0.9 to 10.5 μg L−1 (electrochemical). When compared to other integrated devices based on horizontal designs, the use of a foldable coupling mechanism offered linear response in a lower concentration range and better LOD values for Fe, Ni and Cu. The proposed method successfully measured heavy metals in river water samples with concentrations ranging from 16 to 786 μg L−1, with recovery studies ranging from 76 to 121%. The new method also showed good correlation with conventional atomic absorption spectroscopic methods (95% significance level). Thus, the integration of μPADs and ePADs by a vertical folding mechanism was efficient for multiplexed heavy metal analysis and could be exploited for environmental monitoring.
- This article is part of the themed collection: 150th Anniversary Collection: Electrochemistry and Electroanalytical Approaches


 Please wait while we load your content...
Please wait while we load your content...