Vapochromism induced by intermolecular electron transfer coupled with hydrogen-bond formation in zinc dithiolene complex†
Abstract
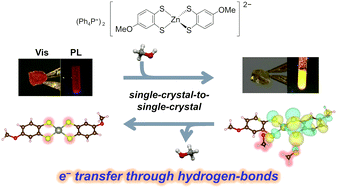
Hydrogen bonding (H-bonding) has long been used for structural regulations. Recently, some examples have been reported where electrical conductivity and magnetism were switched based on direct modulation of electronic states through H-bond. If the application of this strategy is realized to a wide range of functionalities, including optical properties, it will greatly expand the design criteria of functional molecular materials. Here, we report a novel zinc dithiolene complex crystal, which exhibits a new vapochromism induced by electron transfer through H-bonds. This crystal exhibits changes in both visible-light absorption and photoluminescence properties with single-crystal-to-single-crystal (SCSC) transformations caused by methanol or water vapor absorption. Single-crystal structural analysis revealed that the zinc dithiolene complex formed H-bonds with the vapor molecules. Importantly, first-principles calculations based on the periodic crystal structures indicated that the vapochromism in this crystal was solely induced by H-bond formation, not by structural changes. Detailed investigations of the energies and shapes of the crystal orbitals as well as Mulliken population analysis revealed that electron transfer through the H-bonds caused vapochromism in this crystal. The findings provide important insights into the material designs of novel vapochromic materials as well as other stimuli-responsive materials considering the intermolecular H-bonds in the solid-state.


 Please wait while we load your content...
Please wait while we load your content...