High-performing D–π–A–π–D benzothiadiazole-based hybrid local and charge-transfer emitters in solution-processed OLEDs†
Abstract
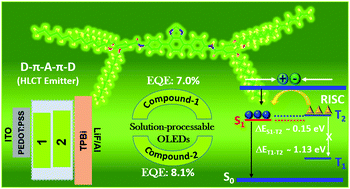
Transforming triplet excitons into singlet excitons is a topic of great interest to material scientists in order to surpass the exciton utilization efficiency (EUE) limit of 25% in electro-fluorescent devices. Towards this goal, the donor–acceptor strategy of molecular design has been proven to be an effective approach to obtain high electroluminescence (EL) efficiency. In this context, herein, we report two benzothiadiazole (BTD)-based donor–acceptor π-conjugated fluorescent molecules, 1 and 2, which exhibited an external quantum efficiency (EQE) as high as 7.0% and 8.1%, respectively, in solution-processed doped green OLED devices. The observed high photoluminescence (PL) quantum yield of 81% for 1 and 85% for 2 corroborate the high EUE values of 43% and 48%, respectively, exceeding the traditional limit (25%), hence suggest the utilization of triplet excitons. The reported emitters combine two parameters, i.e. high PL efficiency and high EUE, which are of key importance to harvest maximum EL in OLED devices. Based on the photophysical (solvatochromic experiment) and quantum chemical calculations, the impacts of the D–π–A–π–D molecular design on the regulation of the locally excited and charge transfer components were revealed, which explained the observed high EQE (>5%) and EUE (>25%) values for the BTD-based emitters. The development of emitters with a hybrid local and charge transfer state in combination with the ‘hot exciton’ mechanism is an important strategy to produce highly efficient fluorescent-emitter materials. Besides the impressive EL properties of the emitters, the studied compound 1 as a chemosensor showed the selective sensing of metal cation (Fe2+) and anion (I−) together with the staining agent of Hibiscus rosa-sinensis pollen grains.


 Please wait while we load your content...
Please wait while we load your content...