Carbon dot mediated trihybrid formation by reduction of GO and in situ gold nanocluster fabrication: photo-switching behaviour and degradation of chemical warfare agent stimulants†
Abstract
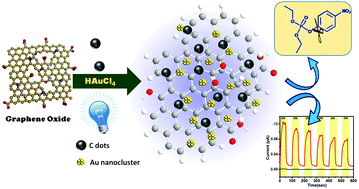
Chemical reduction of graphene oxide (GO) to reduced graphene oxide (rGO) has been regarded as one of the most promising strategies for bulk scale production of graphene-like materials through the restoration of electrical conductivity. In this study, carbon nanodots (C dots) have been used for the reduction of GO in the presence of blue light-emitting diode (LED) irradiation. C dots are also decorated over graphene sheets during the reduction process to make a dihybrid system. If a gold salt is present during the reduction process, gold nanoclusters are formed over the graphene sheet to create a novel trihybrid system consisting of C dots, rGO and Au nanoclusters in one-pot synthesis. The gold nanoclusters on graphene emit red fluorescence and it is very stable. Moreover, both the dihybrid and the trihybrid systems exhibit excellent photo-switching behaviour with a good photocurrent gain. Interestingly, the trihybrid system shows better photocurrent generation than the dihybrid system. Moreover, the trinanohybrid system has been used as an efficient catalyst for the degradation of methylparaoxon (DMNP) showing the potency of the material in the destruction of chemical warfare agents containing phosphoester bonds. This indicates the probable use of this new nanohybrid system with C dots as a good photo-switching material and also a nanocatalyst in future.


 Please wait while we load your content...
Please wait while we load your content...