1,2,3-Triazolyl functionalized thiophene, carbazole and fluorene based A-alt-B type π-conjugated copolymers for the sensitive and selective detection of aqueous and vapor phase nitroaromatics (NACs)†
Abstract
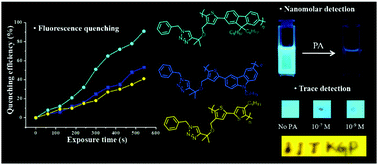
A series of highly emissive π-conjugated A-alt-B type copolymers (P1–P3) appended with a 1,2,3-triazole moiety was synthesized via Suzuki polymerization. The well-defined and soluble π-conjugated copolymers were characterized via multinuclear NMR spectroscopy and tetradetector GPC studies, showing a molecular weight (Mn) in the range of 16.4–20.1 kDa with a polydispersity index in the range of 1.25–1.42. The synthesized emissive π-conjugated polymer probes were explored as fluorescent chemosensors for nitroaromatic compounds (NACs) in solution, vapor and contact mode. Detailed photophysical and sensing studies were performed to understand the polymer–NAC interaction, inducing the selective fluorescence quenching of the π-conjugated polymer probes through the photoinduced electron transfer (PET) mechanism. All the polymeric probes (P1–P3) were highly reversible in nature with NACs, and thus could be reused multiple times. The limit of detection of the probes towards nitroaromatics was found to be in the range of 120–200 ppb with a high association constant in the order of 104 M−1. Furthermore, test paper kits were also fabricated, which allowed the trace detection of picric acid by the naked eye, making it a practical means for the quick, easy and inexpensive on-site detection of NAC-based explosives.


 Please wait while we load your content...
Please wait while we load your content...