Optimization of the carbazole–pyrimidine linking pattern for achieving efficient TADF†
Abstract
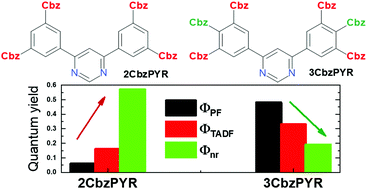
A high fluorescence quantum yield is essential for achieving a high OLED efficiency. Due to their typically strong charge-transfer (CT) character, thermally activated delayed fluorescence (TADF) compounds usually have a lower prompt fluorescence efficiency, and therefore the boosting of the total emission yield (ΦPL) mostly relies on the enhancement of the delayed fluorescence efficiency. In this paper, we present a less frequently used approach for achieving a high ΦPL by maximising both prompt and delayed emission yields of carbazole–pyrimidine compounds. Strong decoupling of HOMO and LUMO and the subsequent emergence of TADF were achieved by introducing four carbazole units at both meta positions of the phenyl unit, though ΦPL was low due to the prominent nonradiative triplet decay rate. Crowding the relatively loose carbazole units together at the para position and both meta positions, conversely, was shown to diminish the nonradiative decay, enhance the radiative decay rate and subsequently boost the fluorescence yield. Efficient blue electroluminescence with a nearly 20% yield was demonstrated for the optimized compound.


 Please wait while we load your content...
Please wait while we load your content...