From Ag2S to luminescent Ag–In–S nanocrystals via an ultrasonic method – an in situ synthesis study in an NMR tube†
Abstract
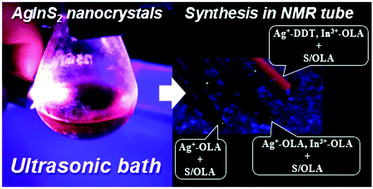
Starting from simple metal precursors (AgNO3 and InCl3), ligands (oleylamine (OLA) and 1-dodecanethiol (DDT)) and popular sulfur precursor S/OLA, we have developed a new method for the preparation of colloidal Ag2S nanocrystals and highly luminescent ternary AgInS2 nanocrystals. The significant advantage of this method is that the nanocrystals can be prepared at room temperature in an ultrasonic bath, without the need for an inert gas atmosphere. In particular, using this method, it was possible to perform the synthesis of the studied nanocrystals in an NMR tube providing direct analytical access to chemical transformations occurring in the reaction mixture during the nanocrystal synthesis. Detailed analysis of the evolution of NMR signals originating from ligands (OLA and DDT) enabled us to identify the nucleation and growth stages as well as the stabilization stage of the nanocrystal formation i.e. the stage in which the ligands were permanently attached to the nanocrystal surface. We also demonstrated that under the analyzed conditions, the reactivities of silver and indium precursors resulting in appropriate cation-to-ligand connections (Ag+–DDT and In3+–OLA) were crucial for the nucleation of ternary AgInS2 nanocrystals exhibiting intense red luminescence.


 Please wait while we load your content...
Please wait while we load your content...