A highly fluorescent covalent organic framework as a hydrogen chloride sensor: roles of Schiff base bonding and π-stacking†
Abstract
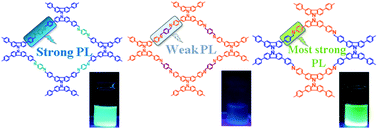
In this paper we report the extremely crystalline structures, high thermal stabilities, and strong fluorescence emissions of covalent organic frameworks (COFs) based on linked carbazole units. We have synthesized three stable luminescent carbazole-linked COFs, namely, BCTB–PD, BCTA–TP, and BCTB–BCTA, through Schiff base condensations of 4,4′,4′′,4′′′-([9,9′-bicarbazole]-3,3′,6,6′-tetrayl)tetrabenzaldehyde (BCTB-4CHO) with p-phenylenediamine (PD), of 4,4′,4′′,4′′′-([9,9′-bicarbazole]-3,3′,6,6′-tetrayl)tetraaniline (BCTA-4NH2) with terephthalaldehyde (TP), and of BCTB-4CHO with BCTA-4NH2, respectively. These COFs had large Brunauer–Emmett–Teller surface areas (up to 2212 m2 g−1) and outstanding thermal stabilities (decomposition temperatures of up to 566 °C). Interestingly, the intramolecular charge transfer (ICT) and fluorescence properties of these COFs were strongly influenced by their types of Schiff base bonding (BCTB-4CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N or BCTA-4N
N or BCTA-4N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH) and the degrees of π-stacking between their COF layers. For example, ICT from the electron-donating carbazole group to the acceptor through the Schiff base units of the type BCTB-4CH
CH) and the degrees of π-stacking between their COF layers. For example, ICT from the electron-donating carbazole group to the acceptor through the Schiff base units of the type BCTB-4CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and increasing the π-stacking distance enhanced the fluorescence emission from the COF. Moreover, BCTB–BCTA, the most fluorescent of our three COFs, functioned as a fluorescent chemosensor for HCl in solution, with outstanding sensitivity and a rapid response.
N and increasing the π-stacking distance enhanced the fluorescence emission from the COF. Moreover, BCTB–BCTA, the most fluorescent of our three COFs, functioned as a fluorescent chemosensor for HCl in solution, with outstanding sensitivity and a rapid response.
- This article is part of the themed collections: Journal of Materials Chemistry C Lunar New Year collection 2021 and 2020 Journal of Materials Chemistry C most popular articles


 Please wait while we load your content...
Please wait while we load your content...