Synthesis of nonlinear optical chromophores with isophorone-derived bridges for enhanced thermal stability and electro-optic activity†
Abstract
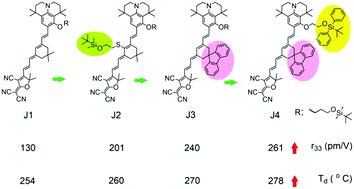
In this work, we developed a series of nonlinear optical chromophores J1–J4 with the same julolidinyl-based donor and tricyanovinyldihydrofuran acceptor with different modified isophorone-derived bridges. The chromophores J2 and J3–J4 were modified with a silane group and a fluorene site-isolation group, respectively, on the bridge leading to enhanced poling efficiency and increased thermal stability. Further modification of chromophore J4 including a tert-butyl(methyl)diphenylsilane group on the donor effectively reduced intermolecular dipole–dipole interactions. Although density functional theory calculations suggested that the four chromophores have similar hyperpolarizabilities, polymeric thin films doped with chromophores J1–J4 have been poled to afford different r33 values of 143, 201, 240, and 261 pm V−1 at 1.31 μm, respectively. The normalized r33 value of chromophore J4 was increased to 14.50 × 10−19 pm cc per (V molecules), which is almost three times higher than that of chromophore J1. In addition, the thermal decomposition temperature of fluorene-containing chromophore J4 was 24 °C higher than that of chromophore J1. All these provide valuable insights into the impact of the isolation group on increased EO behavior and improved thermal stability of the chromophores.


 Please wait while we load your content...
Please wait while we load your content...