Crystalline phase-controlled synthesis of regular and stable endotaxial cesium lead halide nanocrystals†
Abstract
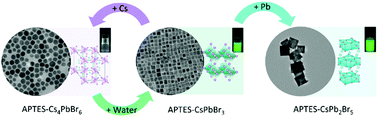
All inorganic lead halide CsPbBr3 perovskite nanocrystals (NCs) have been studied widely due to the tunablility of size, shape and emission along with superior optoelectronic properties. In particular, derivations such as hexagonal Cs4PbBr6 and tetragonal CsPb2Br5 NCs possess improved stability in harsh environments while maintaining unique properties. They also play an essential role in the determination of the phase of the final products during the synthesis of NCs. However, on the one hand, it is still challenging to control the transformation between different perovskite NCs, and subsequently obtain pure phases of each type of NCs. On the other hand, there remains a dispute whether the green fluorescence originates from the pristine CsPb2Br5 NCs or the remaining part of CsPbBr3 NCs. Herein we report a facile and robust control of phase transformation among CsPbBr3, Cs4PbBr6 and CsPb2Br5 NCs, through ligand engineering hot-injection synthesis based on (3-aminopropyl)triethoxysilane (APTES). It reveals rudimentary factors in deciding the structure of the as-synthesized NCs. We successfully achieved the synthesis of the common endotaxial perovskite NCs, Cs4PbBr6 and CsPb2Br5, transformed from cubic CsPbBr3 by precisely tuning the amount of precursors. We found that Cs4PbBr6 NCs exhibit a sharp individual absorption peak in the ultraviolet region; and CsPb2Br5 NCs exhibit 60 nm-thick nanosheet structures with green photoluminescence. Our work realizes the synthesis of nanocrystals of different phase structures and paves ways for further applications such as light-emitting devices and ultra narrow band ultraviolet photodetectors.


 Please wait while we load your content...
Please wait while we load your content...