Iron-coordinating π-conjugated semiconducting polymer: morphology and charge transport in organic field-effect transistors†
Abstract
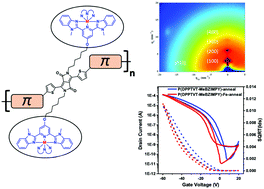
A rational side-chain engineering was performed on a semicrystalline semiconducting conjugated polymer based on diketopyrrolopyrrole (DPP) to incorporate metal-chelating moieties without disruption of the π-conjugation. Specifically, a benzimidazole pyridine-based pincer ligand (MeBZIMPY), known to easily coordinate with M(II) metal centers, was inserted into the conjugated polymer and the effect of Fe(II) coordination on the optical properties was probed by absorption spectroscopy. Moreover, a careful analysis was performed on the metallopolymer in the solid state by grazing incidence X-ray diffraction and atomic force microscopy to evaluate the effect of coordination on the nanoscale morphology and chain packing. Coordination of Fe was directly confirmed by X-ray fluorescence spectroscopy and X-ray near edge absorption spectroscopy experiments. The resulting semiconducting metallopolymer was shown to possess a good solubility in common organic solvents after complexation and a smooth morphology at the nanoscale. The electronic properties of the new metallopolymer were evaluated through fabrication of organic field-effect transistors (OFETs). Interestingly, coordination of Fe(II) by the pincer ligand-containing DPP polymer was shown to enhance the charge mobility in organic field-effect transistors, going from 0.84 to 1.18 cm2 V−1 s−1 upon coordination, which can be directly attributed to the influence of the metal complex on the solid-state morphology. The direct coordination of Fe(II) through pincer ligands is a promising strategy to enhance the charge transport and modulate the optoelectronic properties of π-conjugated polymers without the use of π-conjugation breaking units and/or multi-step synthesis.


 Please wait while we load your content...
Please wait while we load your content...