Determination of phase-formation of (Mg1−xMnx)2Al4Si5O18 (x = 0–1) cordierite solid-solutions via crystallographic sites and luminescence dynamics of Mn2+ centers†
Abstract
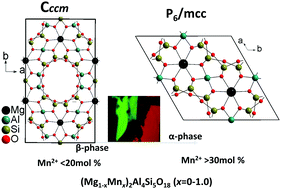
Aluminosilicate with the cordierite structure (Mg2Al4Si5O18) represents a big family of technologically important compounds with typical α- and β-phases. Crystallographic sites for transition metal and rare earth ions in the cordierite structure have always been a controversial topic. In this work, the (Mg1−xMnx)2Al4Si5O18 (x = 0–1.0) solid solution is firstly confirmed via Rietveld structural refinements and luminescence dynamics. The well-crystallized ceramics were prepared by high-temperature solid-state reaction, and then characterized using structural, morphological, luminescence, decays, and thermal quenching measurements. (Mg1−xMnx)2Al4Si5O18 (x = 0–1.0) undergoes structural changes from the orthorhombic β-phase (x = 0–0.2), α-phase (x = 0.3–0.9) and Mn-cordierite (x = 0.95–1.0) (isostructural to β-phase). It is technically meaningful that high-temperature α-phase cordierite Mg2Al4Si5O18 can be stabilized via Mn2+-doping (30–90 mol%). The crystallographic site-occupation of Mn2+ activators is clearly identified. Interestingly, (Mg1−xMnx)2Al4Si5O18 (x = 0.01–1.0) presents tunable colors produced by two distinct luminescence centers, that is, green Mn2+(A) and red Mn2+(B) bands centered at about 530 and 650 nm, respectively. The most efficient excitation wavelengths and decay times of Mn2+(A) are distinct from those of Mn2+(B). The Mn2+(A) green centers are only observed in β-phase cordierite (x = 0.01–0.2) with a dominant emission intensity compared with Mn2+(B), while the Mn2+(B) red centers are observed in all samples (x = 0.01–1.0). The Mn2+(A) center fills in the hexagonal channels, while Mn2+(B) is related to the substitution of Mn2+ for the octahedral Mg2+ site in the cordierite lattices. The luminescence mechanism of Mn2+ in (Mg1−xMnx)2Al4Si5O18 was proposed. The results can be used for basic and application research studies in a wide family of rare earth or transition-metal-doped cordierite materials.


 Please wait while we load your content...
Please wait while we load your content...