Evolution of different defect clusters in Eu3+ doped KMgF3 and Eu3+, Li+ co-doped KMgF3 compounds and the immediate impact on the phosphor characteristics†
Abstract
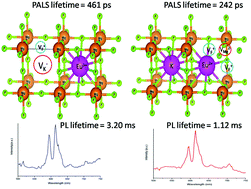
Defects in the lattice have both positive and negative implications for the optical properties of light emitting materials and a great effort is required to understand the defect structure surrounding the activator ion in phosphor materials. The fluoride based perovskite class of compounds such as KMgF3 is always associated with cationic and anionic vacancies, which may give rise to many interesting optical properties. They may also play an important role in determining the phosphor characteristics, when an activator such as Eu3+ ions is doped in KMgF3. This is because defects may alter the local structure of Eu3+ ions, whose characteristic transitions are very sensitive to the local environment. In the first attempt of its kind, we have tried to present a complete picture of the defect structures both in undoped and Li+, Eu3+ ion doped KMgF3 compounds and their influence on the phosphor characteristics. By analysing the changes in the defect structure due to changes in the dopant ions using positron annihilation lifetime spectroscopy supported by EPR and theoretical calculations, it was observed that different defect clusters are being evolved upon changing the dopant ions from Eu3+ to Li+ as co-dopant ions. These changes have a direct impact on the emission characteristics, especially the colour can be tuned to a more deep red region. Using time resolved spectroscopy two different Eu3+ components adjacent to different defect clusters are isolated. The clusters due to K-vacancies were found to change their configuration upon co-doping Li+ ions; VC1 for Eu3+ doped and VC2 for Eu3+ and Li+ co-doped compounds. From the relative percentage of these cluster vacancies, it was possible to make a conclusion about the distribution of Eu3+ and Li+ ions among the different unit cells of KMgF3, which have different defect structures. The study will be helpful in tailoring the defect structure while designing an efficient phosphor material.


 Please wait while we load your content...
Please wait while we load your content...