A novel C6N2 monolayer as a potential material for charge-controlled CO2 capture†
Abstract
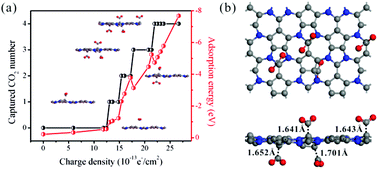
Nowadays, carbon emissions are becoming more and more serious, and therefore it is essential to explore advanced materials with high selectivity and simple operation for capturing and sequestrating CO2. Hence, a new C6N2 monolayer is explored by using the particle swarm optimization (PSO) algorithm for structural search, and then the ability of the charge-controlled C6N2 monolayer to capture CO2 is investigated by the first principles simulation. The results show that the binding strength of CO2 on negatively charged C6N2 can be significantly increased by injecting extra electrons. The process of CO2 capture and release on negatively charged C6N2 is reversible, and thus it can be easily controlled by turning on and off the charges carried. More importantly, the CO2 capture capacity of C6N2 is 5.00 mmol g−1, which is better than 2.90 mmol g−1 of N-doped carbons under 1 atm at 25 °C. In addition, the negatively charged C6N2 monolayer is highly selective for the separation of CO2 from CH4, H2, and N2 mixtures. Based on these remarkable results, C6N2 is a promising charge-controlled CO2 capture material with reversibility, high capacity and selectivity.


 Please wait while we load your content...
Please wait while we load your content...