Tetraphenylethylene-incorporated squaraine dyes: structural and theoretical insights into the diverse emission behaviors in solution and solid state†
Abstract
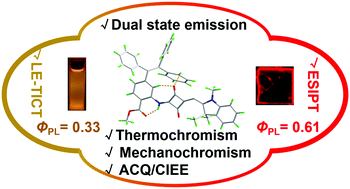
Endowing organic dyes with desired emissive behaviors in diverse states is of great importance for broad practical applications, but it remains a huge challenge. In this study, based on the incorporation of tetraphenylethylene (TPE) into squaraine dyes, we successfully synthesized 2TPE-SQ and TPE-SQ and both exhibited dual state emission with emission efficiencies (ΦPL) up to 0.33 and 0.75, respectively. The intense emission in the crystalline state can be ascribed to highly restricted π–π stacking, while the different degrees of intramolecular phenyl motions in 2TPE-SQ and TPE-SQ result in weak and moderate emissions in solution. Combining density functional theory (DFT) calculations, local excited (LE)/twisted intramolecular charge transfer (TICT) and excited-state intramolecular proton transfer (ESIPT) mechanisms were proposed and accounted well for their very different emission wavelengths in solution and crystalline state, respectively. Besides, other emission behaviors such as aggregation-caused quenching (ACQ), crystalline-induced emission enhancement (CIEE) and thermo-/mechanochromism of these two derivatives were verified in detail, and all these results would bring a new insight into the investigation of organic luminogens in solution and solid state.


 Please wait while we load your content...
Please wait while we load your content...