Atomic defects in monolayer ordered double transition metal carbide (Mo2TiC2Tx) MXene and CO2 adsorption†
Abstract
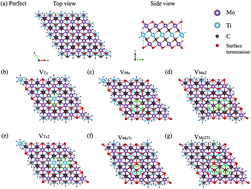
Transition metal carbides (MXenes) with formula Mn+1CnTx (n = 2 and 3) have been emerging as a new family of two-dimensional (2D) materials that have great potential in electronic applications and CO2 conversion catalysts. It has been already found that the electronic and electrochemical properties of Ti3C2Tx MXenes can be tuned by replacing the two outer titanium layers with molybdenum layers. Similar to other 2D materials, intrinsic defects can be formed in the synthesized MXene flakes and the formation of defects can influence the performance of these materials. Herein, we systematically study the effect of the different types of structural defects on the structural stability, electronic behavior, and electrochemical properties of ordered Mo2TiC2Tx terminated with the specific surface functional groups of fluorine, oxygen, and hydroxide. The calculated defect formation energies imply that the formation of defects is dependent on the surface terminations, where the O-terminated MXenes demand more energy than the F- and OH-terminated MXenes. We found that defect formation is more feasible in the outer molybdenum layers than in the inner titanium layer. Our results predicted that the CO2 molecule adsorbs on the defective surfaces through a spontaneous and exothermic process that is critical to its capture, while the perfect surface weakly attracts the molecule through a nonspontaneous and endothermic process. Thus, our study predicts that the electronic and electrochemical properties of Mo2TiC2Tx can be tuned by forming specific defects and these MXenes could be promising materials for CO2 adsorption and conversion.
- This article is part of the themed collection: 2020 Journal of Materials Chemistry C most popular articles


 Please wait while we load your content...
Please wait while we load your content...