Aggregation-induced red-shifted emission and fluorescent patterning of poly(aryleneethynylene) with a lateral AIEgen substituent†
Abstract
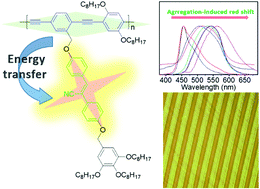
Poly(aryleneethynylene)-based conjugated polymers have attracted significant interest for the fundamental understanding of aggregation behavior, fluorescence emission and electronic coupling through noncovalent stacking interactions. Herein, we investigated the aggregation-induced red-shift in emission of a recently synthesized conjugated polymer P1 with AIEgen substituents on the side chain, and a similar counterpart P2 with the same backbone was also prepared to study the electronic coupling processes and aggregation behaviors from the photophysical properties. The result from 2D-WAXD indicated that P1 had a sheet-like structure and the smectic phase was observed to present interdigitated molecular packing along the smectic layer. The photoluminescence emission showed a distinct red shift of about 110 nm from 454 nm in THF solution to 564 nm in the solid state, which is due to the rotation of the main chain and the stacking behavior of the side-chain substituents. The poly(aryleneethynylene) derivatives were easy to crystallize because of their high degree of rigid conjugation; herein, the introduction of multi-alkyl tails into the terminal of the side chains improved the rheological properties and made them suitable for fabricating highly-ordered fluorescent patterns with high resolution by nanoimprinting lithography, which would open an avenue to extend the future optoelectronic applications.


 Please wait while we load your content...
Please wait while we load your content...