Microwave-assisted synthesis and characterization of iron oxide microfibers
Abstract
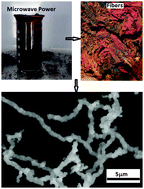
We report a simple, versatile and low-cost method to synthesize iron oxide microfibers with high efficiency and in large quantity. The method is based on the thermal decomposition of iron pentacarbonyl (Fe(CO)5) and silicone oil (SO), and vaporization of carbonyl iron (CI) in a microwave plasma. In this process, the mixture of CI and Fe(CO)5 is brought to a bursting state, and the triggered pressure sprays the reaction products in the form of gas–vapor iron columns. At temperatures lower than that of the iron vapor dew point, the columns freeze, and within tens of seconds microfibers are formed whose dimensions can be controlled by the temperature of the environment or by the microwave field power. SEM images show that their diameters are up to about two microns, arranged in a complex hierarchical structure. Magnetic measurements and EDX elemental analysis show the presence of magnetizable fibers containing iron/oxygen atoms in two proportions. XRD measurements show that the fibers consist of α-Fe2O3, γ-Fe2O3 and Fe3O4 iron oxides. We explain the physical mechanisms underlying microfiber formation by developing a theoretical framework based on the Higbie model. The obtained results can be used for fabrication of composite materials with magnetodielectric properties.


 Please wait while we load your content...
Please wait while we load your content...