A conjugated polyelectrolyte interfacial modifier for high performance near-infrared quantum-dot photodetectors†
Abstract
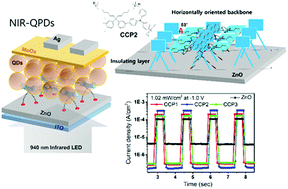
The photodetection properties of near-infrared quantum-dot photodetectors (NIR-QPDs) have been significantly improved by introducing novel cationic conjugated polyelectrolytes (CCPs) on ZnO. The formation of a quaternary ammonium salt at the end of the pendant groups of the CCPs generates a permanent dipole moment on the ZnO surface and effectively modifies the work function of ZnO, improving the charge transport at the interfaces between the ZnO and QD layers. The cationic charge density and the geometrical structures of CCP1–3 had crucial effects in determining the photo-responsivity (R) and detectivity (D*) of the NIR-QPDs. The higher cationic charge density of CCP2 improved the electron accepting properties of ZnO, resulting in 1.6–5.1-fold increased R values under 940 nm IR irradiation (0.1 mW cm−2, −1 V) compared to the ZnO-, ZnO/CCP1-, and ZnO/CCP3-based devices. Notably, the horizontally-aligned backbone of CCP2 toward the substrate gives a uniform film surface and insulating layer on ZnO, which improved the charge transport and maintained a relatively low and constant dark current under negative bias. As a result, D* of the CCP2-treated NIR-QPDs was increased by up to 1880% compared to the pristine ZnO-based devices, and by 200–250% compared to the CCP1- and CCP3-based devices.


 Please wait while we load your content...
Please wait while we load your content...