Near-infrared photothermal liposomal nanoantagonists for amplified cancer photodynamic therapy†
Abstract
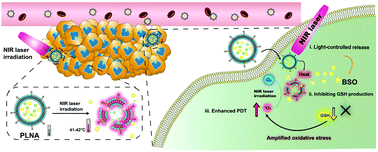
Photodynamic therapy (PDT) has been demonstrated to be a promising strategy for the treatment of cancer, while its therapeutic efficacy is often compromised due to excessive concentrations of glutathione (GSH) as a reactive oxygen species (ROS) scavenger in cancer cells. Herein, we report the development of near-infrared (NIR) photothermal liposomal nanoantagonists (PLNAs) for amplified PDT through through the reduction of intracellular GSH biosynthesis. Such PLNAs were constructed via encapsulating a photosensitizer, indocyanine green (ICG) and a GSH synthesis antagonist, L-buthionine sulfoximine (BSO) into a thermal responsive liposome. Under NIR laser irradiation at 808 nm, PLNAs generate mild heat via a ICG-mediated photothermal conversion effect, which leads to the destruction of thermal responsive liposomes for a controlled release of BSO in a tumor microenvironment, ultimately reducing GSH levels. This amplifies intracellular oxidative stresses and thus synergizes with PDT to afford an enhanced therapeutic efficacy. Both in vitro and in vivo data verify that PLNA-mediated phototherapy has an at least 2-fold higher efficacy in killing cancer cells and inhibiting tumor growth compared to sole PDT. This study thus demonstrates a NIR photothermal drug delivery nanosystem for amplified photomedicine.


 Please wait while we load your content...
Please wait while we load your content...