Supramolecular magnetonanohybrids for multimodal targeted therapy of triple-negative breast cancer cells†
Abstract
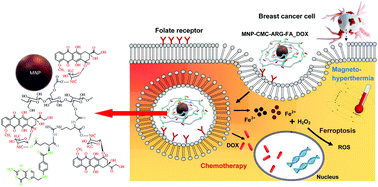
Despite the undeniable advances in recent decades, cancer remains one of the deadliest diseases of the current millennium, where the triple-negative breast cancer (TNBC) is very aggressive, extremely metastatic, and resistant to conventional chemotherapy. The nanotheranostic approach focusing on targeting membrane receptors often expressed at abnormal levels by cancer cells can be a strategic weapon for fighting malignant tumors. Herein, we introduced a novel “all-in-one nanosoldier” made of colloidal hybrid nanostructures, which were designed for simultaneously targeting, imaging, and killing TNBC cells. These nanohybrids comprised four distinct components: (a) superparamagnetic iron oxide nanoparticles, as bi-functional nanomaterials for inducing ferroptosis via inorganic nanozyme-mediated catalysis and magnetotherapy by hyperthermia treatment; (b) carboxymethyl cellulose biopolymer, as a water-soluble capping macromolecule; (c) folic acid, as the membranotopic vector for targeting folate receptors; (d) and doxorubicin (DOX) drug for chemotherapy. The results demonstrated that this novel strategy was highly effective for targeting and killing TNBC cells in vitro, expressing high levels of folate membrane-receptors. The results evidenced that three integrated mechanisms triggered the deaths of the cancer cells in vitro: (a) ferroptosis, by magnetite nanoparticles inducing a Fenton-like reaction; (b) magneto-hyperthermia effect by generating heat under an alternate magnetic field; and (c) chemotherapy, through the DOX intracellular release causing DNA dysfunction. This “all-in-one nanosoldier” strategy offers a vast realm of prospective alternatives for attacking cancer cells, combining multimodal therapy and the delivery of therapeutic agents to diseased sites and preserving healthy cells, which is one of the most critical clinical challenges faced in fighting drug-resistant breast cancers.
- This article is part of the themed collection: Celebrating Latin American Talent in Chemistry


 Please wait while we load your content...
Please wait while we load your content...