pH-Responsive nanocomposite fibres allowing MRI monitoring of drug release†
Abstract
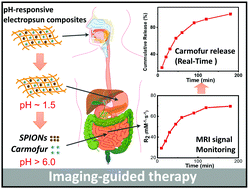
Magnetic resonance imaging (MRI) is one of the most widely-used non-invasive clinical imaging tools, producing detailed anatomical images whilst avoiding side effects such as trauma or X-ray radiation exposure. In this article, a new approach to non-invasive monitoring of drug release from a delivery vehicle via MRI was developed, using pH-responsive Eudragit L100 and S100 fibres encapsulating superparamagnetic iron oxide nanoparticles (SPIONs) and carmofur (a drug used in the treatment of colon cancer). Fibres were prepared by electrospinning, and found to be smooth and cylindrical with diameters of 645 ± 225 nm for L100 and 454 ± 133 nm for S100. The fibres exhibited pH responsive dissolution behaviour. Around the physiological pH range, clear pH-responsive proton relaxation rate changes due to matrix swelling/dissolution can be observed: r2 values of L100 fibres increase from 29.3 ± 8.3 to 69.8 ± 2.5 mM−1 s−1 over 3 h immersion in a pH 7.4 medium, and from 13.5 ± 2.0 mM−1 s−1 to 42.1 ± 3.0 mM−1 s−1 at pH 6.5. The r2 values of S100 fibres grow from 30.4 ± 4.4 to 64.7 ± 1.0 mM−1 s−1 at pH 7.4, but at pH 6.5, where the S100 fibres are not soluble, r2 remains very low (< 4 mM−1 s−1). These dramatic changes in relaxivity demonstrate that pH-responsive dissolution results in SPION release. In vitro drug release studies showed the formulations gave rapid release of carmofur at physiological pH values (pH 6.5 and 7.4), and acid stability studies revealed that they can protect the SPIONs from digestion in acid environments, giving the fibres potential for oral administration. Exploration of the relationship between relaxivity and carmofur release suggests a linear correlation (R2 > 0.94) between the two. Mathematical equations were developed to predict carmofur release in vitro, with very similar experimental and predicted release profiles obtained. Therefore, the formulations developed herein have the potential to be used for non-invasive monitoring of drug release in vivo, and could ultimately result in dramatic reductions to off-target side effects from interventions such as chemotherapy.
- This article is part of the themed collection: 2023 Journal of Materials Chemistry Lectureship shortlisted candidates


 Please wait while we load your content...
Please wait while we load your content...