Self-recognizing and stimulus-responsive carrier-free metal-coordinated nanotheranostics for magnetic resonance/photoacoustic/fluorescence imaging-guided synergistic photo-chemotherapy†
Abstract
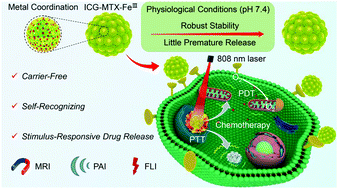
Carrier-free nanotheranostics directly assembled by using clinically used photosensitizers and chemotherapeutic drugs are a promising alternative to tumor theranostics. However, the weak interaction-driven assembly still suffers from low structural stability against disintegration, lack of targeting specificity, and poor stimulus-responsive property. Moreover, almost all exogenous ligands possess no therapeutic effect. Enlightened by the concept of metal–organic frameworks, we developed a novel self-recognizing metal-coordinated nanotheranostic agent by the coordination-driven co-assembly of photosensitizer indocyanine green (ICG) and chemo-drug methotrexate (MTX, also served as a specific “targeting ligand” towards folate receptors), in which ferric (FeIII) ions acted as a bridge to tightly associate ICG with MTX. Such carrier-free metal-coordinated nanotheranostics with high dual-drug payload (∼94 wt%) not only possessed excellent structural and physiological stability, but also exhibited prolonged blood circulation. In addition, the nanotheranostics could achieve the targeted on-demand drug release by both stimuli of internal lysosomal acidity and external near-infrared laser. More importantly, the nanotheranostics could self-recognize the cancer cells and selectively target the tumors, and therefore they decreased toxicity to normal tissues and organs. Consequently, the nanotheranostics showed strongly synergistic potency for tumor photo-chemotherapy under the precise guidance of magnetic resonance/photoacoustic/fluorescence imaging, thereby achieving highly effective tumor curing efficiency. Considering that ICG and bi-functional MTX are approved by the Food and Drug Administration, and FeIII ions have high biosafety, the self-recognizing and stimulus-responsive carrier-free metal-coordinated nanotheranostics may hold potential applications in tumor theranostics.


 Please wait while we load your content...
Please wait while we load your content...