A tumor-sensitive biological metal–organic complex for drug delivery and cancer therapy†
Abstract
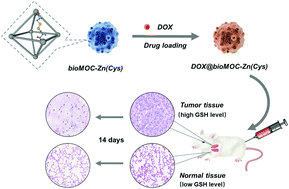
Metal–organic complexes (MOCs) or metal–organic frameworks (MOFs) have attracted increasing interest for constructing nanoscale drug delivery systems for cancer therapy. However, conventional MOC/MOF materials usually contain toxic metals or low-biocompatible organic ligands. Also, current approaches for creating tumor-sensitive nanocarriers are always based on the instability of coordination bonds under acidic conditions, or through post-synthetic modification with sensitive molecules. As a matter of fact, it is more facile to fabricate tumor-sensitive MOCs/MOFs based on the stimuli-responsiveness of organic ligands. In this study, a novel tumor-sensitive biological MOC (bioMOC-Zn(Cys)) was created through the assembly of endogenous Zn2+ ions and the small biological molecule (L-cystine, Cys). The disulfide bond in L-cystine is cleavable by the overexpressed GSH in tumor cells, thus achieving rapid release of drugs from nanocarriers. By encapsulating doxorubicin (DOX) in bioMOC-Zn(Cys), DOX@bioMOC-Zn(Cys) displayed higher cellular uptake and cytotoxicity in cancer cells than free DOX. In vivo investigations indicated that DOX@bioMOC-Zn(Cys) largely inhibited tumor growth and reduced side effects. Remarkably, since both metal ions and organic ligands were obtained from biological sources, bioMOC-Zn(Cys) exhibited superior biocompatibility. This study presents a new method for fabricating MOC-based nanodrugs with high tumor-sensitivity and low toxicity.


 Please wait while we load your content...
Please wait while we load your content...