Enzyme assisted peptide self-assemblies trigger cell adhesion in high density oxime based host gels†
Abstract
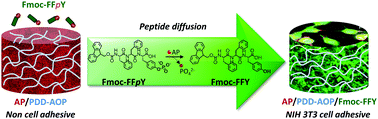
Peptide supramolecular self-assemblies are recognized as important components in responsive hydrogel based materials with applications in tissue engineering and regenerative medicine. Studying the influence of hydrogel matrices on the self-assembly behavior of peptides and interaction with cells is essential to guide the future development of engineered biomaterials. In this contribution, we present a PEG based host hydrogel material generated by oxime click chemistry that shows cellular adhesion behavior in response to enzyme assisted peptide self-assembly (EASA) within the host gel. This hydrogel prepared from poly(dimethylacrylamide-co-diacetoneacrylamide), poly(DMA–DAAM) with high molar fractions (49%) of DAAM and dialkoxyamine PEG cross-linker, was studied in the presence of embedded enzyme alkaline phosphatase (AP) and a non-adhesive cell behavior towards NIH 3T3 fibroblasts was observed. When brought into contact with a Fmoc-FFpY peptide solution (pY: phosphorylated tyrosine), the gel forms intercalated Fmoc-FFY peptide self-assemblies upon diffusion of Fmoc-FFpY into the cross-linked hydrogel network as was confirmed by circular dichroism, fluorescence emission spectroscopy and confocal microscopy. Nevertheless, the mechanical properties do not change significantly after the peptide self-assembly in the host gel. This enzyme assisted peptide self-assembly promotes fibroblast cell adhesion that can be enhanced if Fmoc-F-RGD peptides are added to the pre-gelator Fmoc-FFpY peptide solution. Cell adhesion results mainly from interactions of cells with the non-covalent peptide self-assemblies present in the gel despite the fact that the mechanical properties are very close to those of the native host gel. This result is in contrast to numerous studies which showed that the mechanical properties of a substrate are key parameters of cell adhesion. It opens up the possibility to develop a diverse set of hybrid materials to control cell fate in culture due to tailored self-assemblies of peptides responding to the environment provided by the host guest gel.


 Please wait while we load your content...
Please wait while we load your content...
