A drug delivery system obtained by hot-melt processing of zein plasticized by a pharmaceutically active ionic liquid†
Abstract
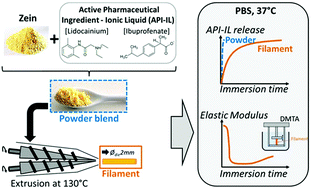
Zein-based filaments containing 20 weight% [Lidocainium][Ibuprofenate] used as a doubly Active Pharmaceutical Ingredient–Ionic Liquid (API–IL) were obtained by extrusion at 130 °C. The plasticizing effect of the active ingredient on the zein amorphous matrix was assessed by differential scanning calorimetry, with a decrease in the glass transition temperature (Tg) from 77 °C, for the raw zein, to 53 °C. After storage under standard conditions (relative humidity 59%, 20 °C) the extrudates were rigid, with a high storage modulus (E′) of about 3 GPa at ambient temperature. They had a main mechanical relaxation (Tα) beginning at 55 °C, leading to their flowing at temperatures above 130 °C, as determined by dynamic mechanical analysis, with E′ below 1 MPa and tan δ above 1. Their structure was evaluated by wide angle X-ray scattering and NMR analysis was used to evaluate the API–IL stability after thermomechanical processing. Release experiments performed under simulated physiological conditions on filaments evidenced a release of 85% after 7 days immersion. These results demonstrate the advantage of using an API–IL as plasticizer of a resorbable biopolymer. The resulting material can be shaped by a continuous thermomechanical process and used as a drug delivery system.


 Please wait while we load your content...
Please wait while we load your content...